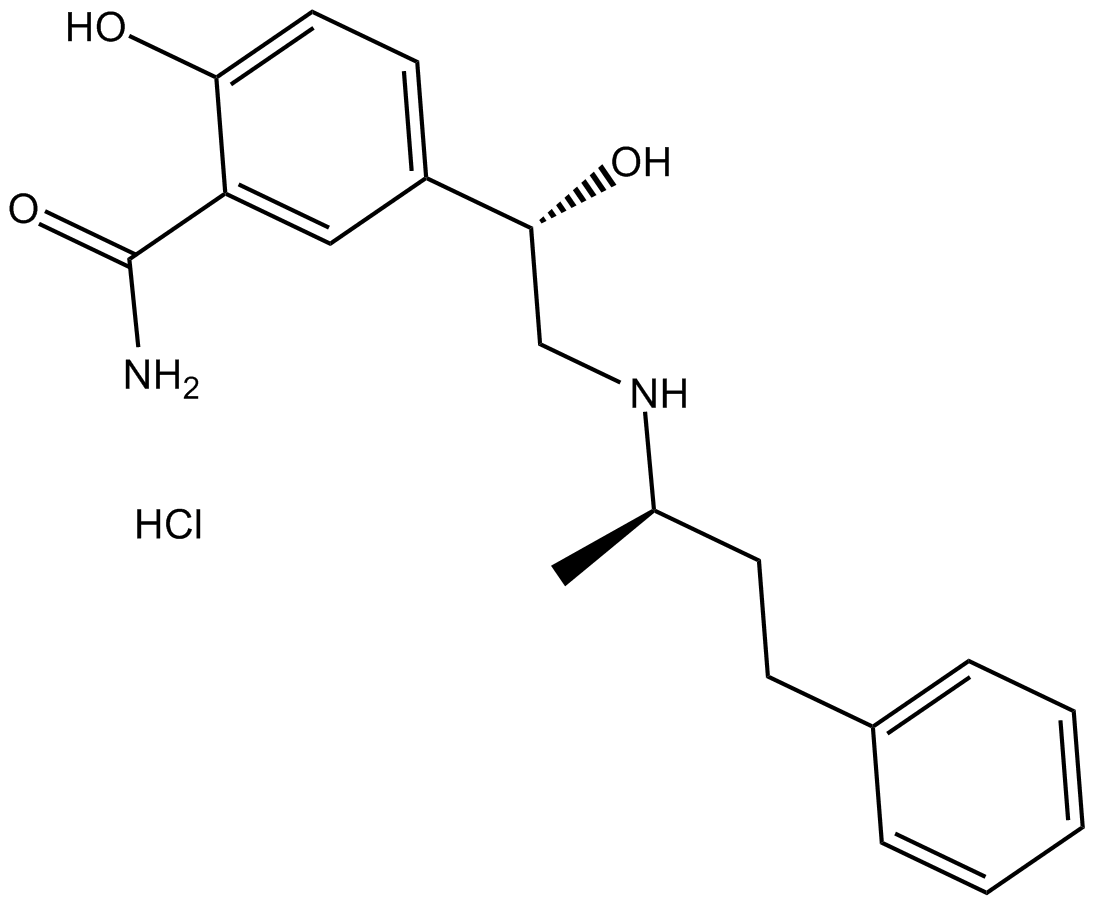
Class 2 Medicines Recall: Tillomed Laboratories Limited, Labetalol 200mg Tablets, EL(23)A/23
Por um escritor misterioso
Descrição
Tillomed Laboratories Limited is recalling one batch of Labetalol 200mg Tablets due to an error on the foil blister packaging.

Labetalol Hydrochloride (Almaject, Inc.): FDA Package Insert, Page 5

Difference between PGS and PGD
:quality(70)/cloudfront-us-east-1.images.arcpublishing.com/cmg/2JJAB32VVFAMHKYQ6NBJVVNDUQ.jpg)
Blood pressure, heart condition medication recalled after labels were switched – KIRO 7 News Seattle

Teva Pharmaceuticals USA Issues Voluntary Nationwide Recall of all Amlodipine/Valsartan Combination Tablets and Amlodipine/Valsartan/Hydrochlorothiazide Combination Tablets that are Within Expiry

Teva Pharmaceuticals announces recall of two blood pressure drugs.

Doctors: Blood pressure drug substitutes are available for patients affected by recalls

Labetalol batch recalled due to wrong dosage labelling - The Pharmacist

Dispensing & Supply Archives - Page 10 of 84 - Community Pharmacy England

LABETALOL HYDROCHLORIDE TABLETS USP Rx only
Blood pressure medication recall: What you need to know - ABC News
de
por adulto (o preço varia de acordo com o tamanho do grupo)